 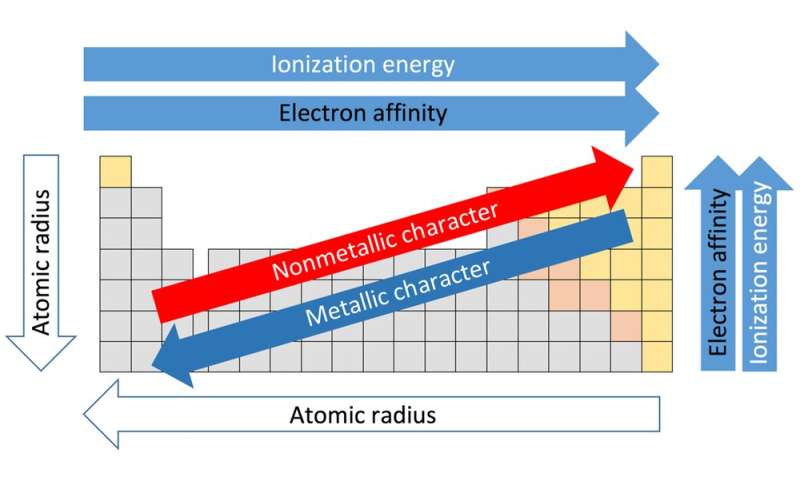
Nonmetals have more valence electrons, which makes it easier for them to gain electrons and complete a set. Nonmetals have more electron affinity than metals do because of their atomic structure. This means that the electron affinity value of nonmetals is typically negative. This is because nonmetals have enough energy to form negatively charged ions, anions. Unlike metals, when a nonmetal gains an electron, the amount of energy change is typically negative. Cesium (Cs) Electron Affinity: 46 KJ mol-1.Rubidium (Rb) Electron Affinity: 47 KJ mol-1.Sodium (Na) Electron Affinity: 53 KJ mol-1.Lithium (Li) Electron Affinity: 60 KJ mol-1.As an example of the fact that metals have low electron affinity, look at the following electron affinity values for the metals found in Group 1 of the periodic table: For this reason, many metals have very low electron affinities.Įlectron affinities are given in kj/mol (joules per mole), a measurement of given energy per amount of material. This is because metals don’t exert a very strong pull on their valence electrons and are therefore lose electrons in the valence shell rather easily, becoming cations. 
The addition of an electron to a metal element requires energy. This means that the energy required overwhelms the energy released by the electron attachment process, and so the second electron affinity will be positive. The ion is now negative, and more energy is necessary when an electron is being added to a negative ion. This is because energy is released when an electron is added to a neutral atom. The initial addition of an electron to a neutral atom, the first electron affinity, will always have negative energy. The electron affinity of an atom depends upon when it is added to the atom. “The protons are positive and the electrons are negative and, of the two, I am sure that the electrons are nicer.” - Robert Benchley It’s important to remember that, so you’ll know that electron affinities deal exclusively with negative ions of atoms and that their use is almost always relegated to the elements found within groups 16 and 17 of the element table. Ionization energies deal with the formation of positive ions while electron affinities deal with the formation of negative ions. The energy of an atom has can be gained or lost through chemical reactions, so these chemical reactions form either anions or cations. Positively charged atoms are called cations while negatively charged ions are called anions. Ions of atoms may have a net positive charge or a net negative charge. In other words, the greater an element’s tendency to gain electrons, the more reactive the element is. Reactivity and electron affinity are tightly correlated, with the reactivity of an element increasing as the electron affinity increases. Elements that have only one electron in their valence shell, like alkali metals, or are missing a single electron in the shell (like halogens) are the most reactive elements. The electrons in the valence shell are known as valence electrons, and they are the electrons most capable of bonding with other atoms to create chemicals bonds and molecules.Įlements that have complete valence shells, like the noble gases, are stable and chemically non-reactive. 
The outermost layer of the electron shells is known as the valence shell, and it’s usually the only layer that matters in chemistry. The electrons orbit the nucleus of the atom, and they orbit in multiple layers known as shells. “We’re protons and electrons/ Residing in one nucleus.” - AfeefaĮlectrons are about 1800 times smaller than either neutrons or protons, and they have a negative charge. The neutrons are used as a point of comparison to find the mass of electrons and protons. The neutrons within the atom have no charge, hence their neutrality. The number of protons within an atom is the element’s atomic number. The protons in the atom are positively charged, and the number of protons found within the nucleus basically defines what element an atom is. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
